

Pseudocapacitors: Working Principles and Applications
Catalog
What is a Pseudocapacitor?Pseudocapacitor DiagramWorking Principle of PseudocapacitorsHow to Connect a Pseudocapacitor?Types of PseudocapacitorsPseudocapacitor vs. SupercapacitorAdvantages of PseudocapacitorsDisadvantages of PseudocapacitorsApplications of PseudocapacitorsIn SummaryRelated ArticlesA supercapacitor is a specialized energy storage device characterized by exceptionally high capacitance, combining the functional properties of conventional capacitors and batteries within a single unit. Compared to standard capacitors, supercapacitors offer significantly higher energy storage; relative to batteries, they deliver superior power output. Also referred to as ultracapacitors, these devices are user-friendly and inherently safe.
Supercapacitors are categorized into three main types to suit diverse applications: Electric Double-Layer Capacitors (EDLCs), pseudocapacitors, and hybrid capacitors. This article focuses on pseudocapacitors—one class of supercapacitors—exploring their operating mechanism and practical applications.
What is a Pseudocapacitor?
Pseudocapacitor Definition
Pseudocapacitors, also known as faradaic supercapacitors, differ fundamentally from EDLCs. Their electrodes incorporate redox-active materials, enabling electrical energy storage through a distinct mechanism compared to EDLCs.

Pseudo Capacitor
In practice, only a portion of the charge storage comes from EDLC effects, while faradaic mechanisms — including redox reactions, electrosorption, and intercalation — enable much higher levels of energy transfer and storage.
When an external potential is applied to the capacitor, rapid and reversible redox reactions take place at the electrode surface, involving charge transfer between the electrode and electrolyte. The charge–discharge mechanism of pseudocapacitors is similar to that of electrochemical batteries.
Pseudocapacitor Diagram
A pseudocapacitor acts as a hybrid between a battery and an EDLC (electric double-layer capacitor). It consists of two electrodes separated by an electrolyte, with charge storage occurring primarily through both chemical and electrostatic processes.
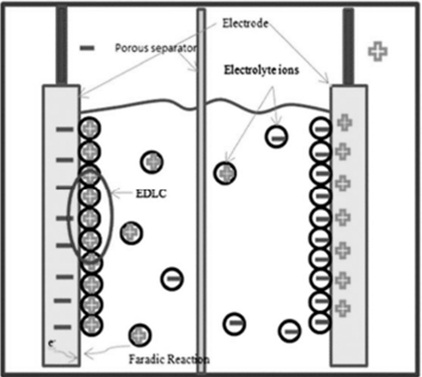
Pseudocapacitor Diagram
The chemical process primarily involves charge transfer via redox (reduction‑oxidation) reactions.
While the charge storage mechanism is similar to that in batteries, charge–transfer rates are much faster in pseudocapacitors, thanks to the thin layer of redox material on the electrode surface. This reduces ion diffusion limitations from the electrolyte into the bulk structure.
Pseudocapacitors achieve higher capacitance values because they utilize multiple charge‑storage mechanisms simultaneously.
Working Principle of Pseudocapacitors
The working principle of a pseudocapacitor involves storing electrical energy through the transfer of electron charge between the electrode and electrolyte. This is achieved via reduction-oxidation (redox) reactions, electrosorption, and intercalation processes—collectively referred to as pseudocapacitance. In an electrochemical capacitor, pseudocapacitors are a key component, working alongside Electric Double-Layer Capacitors (EDLCs) to form a supercapacitor.
Pseudocapacitors are typically composed of metal sulfides, metal oxides, metal hydroxides, metal nitrides, and conducting polymers. Examples of pseudocapacitive materials include metal oxides such as RuO₂, NiO, MnO₂, and Co₃O₄, as well as conducting polymers like polyaniline and polypyrrole.
Energy storage in pseudocapacitors occurs primarily through faradaic reactions, while also incorporating electrostatic charge storage involving charge transfer between the electrode and electrolyte. When a voltage is applied to a pseudocapacitor, both reduction and oxidation reactions take place on the electrode material. The faradaic processes employed in these capacitors enhance electrochemical reactions, resulting in higher specific capacitance and energy densities compared to EDLCs.
Pseudocapacitor materials boost energy density, allowing energy to be stored in the volume of the electrode material as well as on its surface. A key characteristic of these materials is that they are either electrically conductive or capable of existing in two oxidation states within a specific potential window.
Due to their unique charge-storage mechanisms, pseudocapacitors offer the highest capacitance density among all capacitor types. The total electric charge stored via pseudocapacitance is linearly proportional to the applied voltage.
Faradaic Reaction
A faradaic reaction is a chemical reaction where electron transfer leads to oxidation or reduction. This reaction is governed by Faraday’s Law, which states that the amount of chemical reaction induced by an electric current is proportional to the quantity of electricity passed through the system.
Redox Reaction
A redox reaction is a chemical reaction involving the transfer of electrons between two participating reactants. This electron transfer can be easily identified by monitoring changes in the oxidation states of the reacting species.
How to Connect a Pseudocapacitor?
The circuit configuration of a pseudocapacitor is illustrated below. As shown in the diagram, Rs represents the series resistance, while the pseudocapacitance (Cᵩ) depends primarily on the applied potential. RD denotes the Faradaic resistance, which operates during discharge until the ions are depleted, and RF is the electrode-electrolyte resistance. At specific potentials, the pseudocapacitance (Cᵩ) exceeds the double-layer capacitance (Cdl). For this reason, connecting pseudocapacitors in parallel is highly effective for increasing their total capacitance, as this configuration follows the additive law of capacitance.
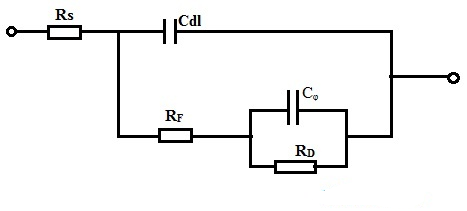
Pseudocapacitor Circuit
Types of Pseudocapacitors
Pseudocapacitors are classified into two main types based on the electrode materials used for charge storage, as outlined below:
Metal Oxides
Metal oxides are a class of pseudocapacitive materials that exhibit fast, reversible redox reactions on the surface of the electrode material. They feature low resistance and high specific capacitance, making them easy to use in constructing high-power supercapacitors.
The most commonly used metal oxides for supercapacitor electrodes include MnO₂, RuO₂, NiO, SnO₂, IrO₂, Fe₃O₄, V₂O₅, Co₂O₃, and MoO. Among these, RuO₂ is regarded as the most capable electrode material for supercapacitor applications due to its high electrical conductivity and specific capacitance. However, its practical use is limited by the scarcity of ruthenium (Ru) on Earth. For this reason, identifying low-cost pseudocapacitive materials is essential.
Metal oxides exhibit different oxidation states at various potentials and possess crystalline structures that enable high conductivity, allowing charges to transfer within their network. They can change their oxidation states, and protons can be inserted into or removed from the oxide lattice during surface oxidation and reduction reactions.
Conducting Polymers
Conducting polymers are widely used in redox pseudocapacitors due to their rapid, reversible oxidation-reduction processes, relatively low cost, and good electrical conductivity.
The most commonly used conducting polymers include PPy (polypyrrole), PANI (polyaniline), PTh (polythiophene), PPV (poly-p-phenylene vinylene), and PEDOT (poly(3,4-ethylenedioxythiophene)). These materials are typically produced through either electrochemical oxidation or chemical polymerization of monomers, and their conductivity is achieved through a conjugated bond system in the polymer backbone.
Compared to carbon-based electrode materials, conducting polymers offer higher capacitance, improved conductivity, and lower equivalent series resistance. During oxidation and reduction reactions, ions can easily migrate from the electrolyte to the conducting polymers and then be released back into the electrolyte.
These polymers exhibit highly reversible behavior, leading to excellent cycling stability because no phase transition occurs. They become positively or negatively charged through redox reactions, which further enhances their conductivity.
Pseudocapacitor vs. Supercapacitor
The key differences between a pseudocapacitor and a supercapacitor are outlined below, along with details on the advantages, disadvantages, and applications of pseudocapacitors.
Key Differences
| Pseudocapacitor | Supercapacitor |
|---|---|
| Also called a faradaic supercapacitor. | Also known as an ultracapacitor or electrochemical capacitor. |
| Available in two types: Metal Oxide and Conducting Polymers. | Available in three types: Electrostatic Double-Layer Capacitors (EDLCs), Pseudocapacitors, and Hybrid Capacitors. |
| Stores energy through both physical and chemical processes. | Relies entirely on physical energy storage (for EDLCs, the primary type). |
| Higher specific capacitance. | Lower specific capacitance. |
| High energy density. | Low energy density. |
| Lower cycle life and stability. | Higher cycle life and stability. |
| Relies on redox reactions for energy storage. | Does not rely on redox reactions (for EDLCs). |
| High power density. | Very high power density. |
| Medium cost per unit of energy. | High cost per unit of energy. |
Advantages of Pseudocapacitors
- High power density.
- Significantly longer service life compared to many traditional energy storage devices.
- Much faster charging and discharging speeds than lithium-ion batteries.
- Pseudocapacitor materials enhance energy density, enabling energy storage both in the bulk of the electrode materials and on their surface.
Disadvantages of Pseudocapacitors
- Lower energy density than batteries, making them unsuitable as a direct replacement for batteries in energy storage applications.
- Not ideal for long-term energy storage devices.
- Output voltage decreases linearly as the capacitor discharges.
Applications of Pseudocapacitors
- Store electrical energy through faradaic reactions.
- Serve as a key component of electrochemical capacitors, working with EDLCs to form supercapacitors.
- Used in consumer electronics.
- Integrated into wearable and flexible electronics.
- Applied in regenerative braking systems for automobiles.
- Used in kinetic energy (KE) recovery systems, such as those in cranes, elevators, and wind turbines.
In Summary
In summary, this provides a concise overview of pseudocapacitors, their working principle, and their applications. While EDLC materials cannot sufficiently increase energy density, pseudocapacitor materials enhance energy density and enable energy storage both in the volume of electrode materials and on their surface. The main advantages of pseudocapacitors include cost-effectiveness, light weight, eco-friendliness, flexibility, improved safety, and tunable electrochemical properties. A common question: What is an EDLC?
Related Articles
What is a Manual Transfer Switch: Working Principle & Applications
What is a Membrane Switch? Working Principle and Applications
Subscribe to JMBom Electronics !













